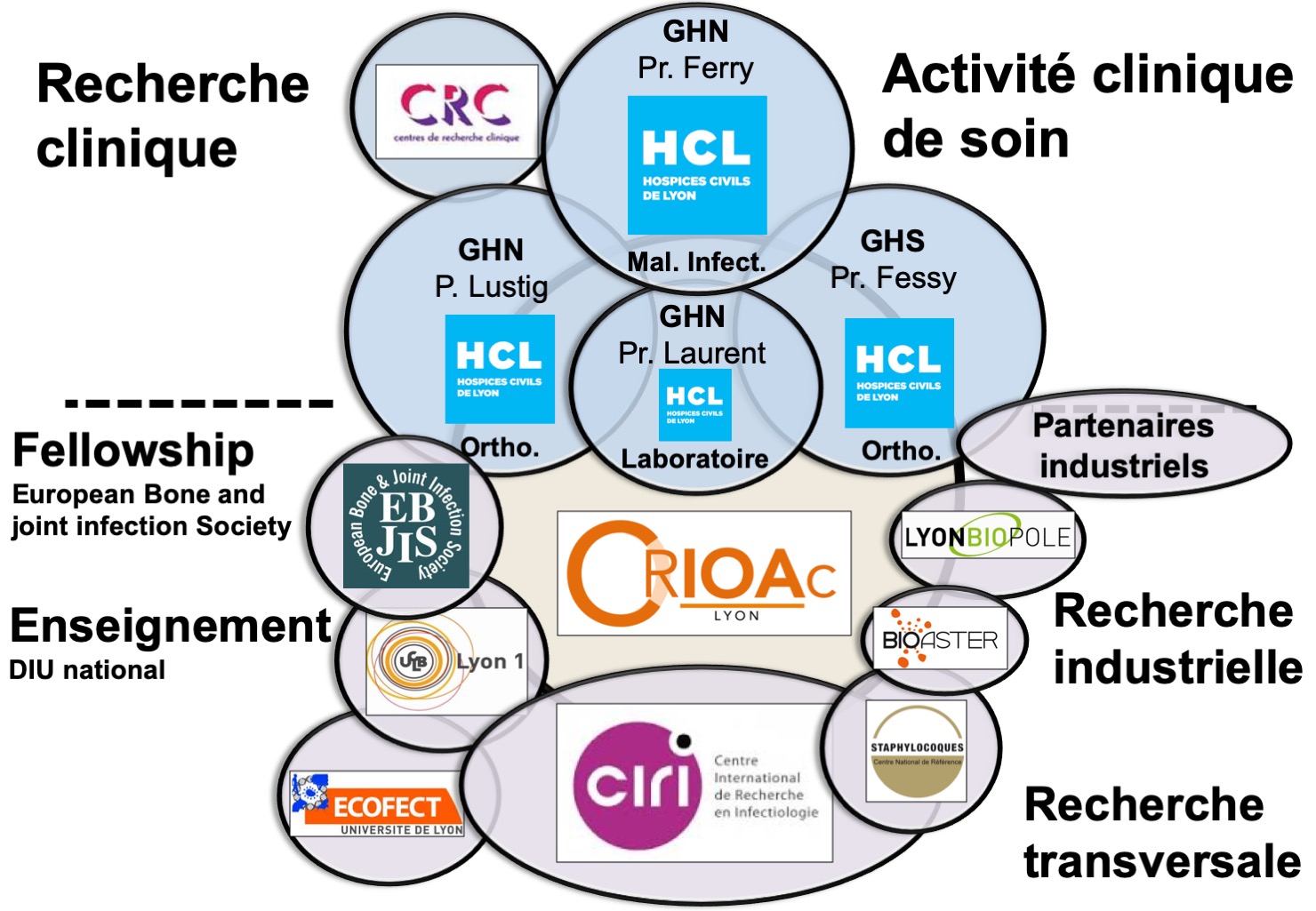
Clinical Research
In the field of BJI, many unknowns remain with respect to therapeutics. The different strategies, types of implants, types of cement, and the different commonly used antibiotics need to be evaluated. New coated implants, limiting the risk of superinfection, are available. Cements, locally diffusing 1 ou 2 antibiotics, can be used. Bone substitutes that release high concentrations of antibiotics are also now available. These new devices are expensive, but they are difficult to evaluate. They could mean a real clinical benefit for patients, but their exact place in the therapeutic arsenal is not yet known. During a BJI, it is sometimes necessary to use antibiotics which do not have market authorisation (AMM, in France). Antibiotics which have market authorisation for other indications, are available in France, but have not been evaluated as a possible treatment for BJI. Due to intolerance or resistance to the established treatment, we are frequently led to use these, often expensive, antibiotics, in response to BJI. The evaluation of the use of off-label antibiotics is crucial in order to judge tolerance and effectiveness. This this end, we have published a number of articles describing the efficacy and safety of non-approved antibiotics in CRIOAc patients.

Le CRIOAc Lyon also works with the Centre of Clinical Research (CRC) with Pr. Fabien Zoulim, as its scientific head. The hospital-based CRC is backed by the Ministry of Health. It facilites clinical research with the support of the HCL Department of Innovation and Clinical Research Direction de la Recherche Clinique et de l’Innovation des HCL. The CRC supports medical Principal Investigators of either academic or industry-sponsored studies. The CRC provides assistance at all stages of the clinical research protocol : set-up, patient recruitment, data acquisition and logistics. The CRC connects the various actors of clinical research : patients, clinicians and institutional or industial partners. The CRC has participated in 100-150 studies per year and has enrolled 2000 to 3000 patients per year. Most of these studies were industry-sponsored, on subjects such as viral hepatitis or HIV. Thanks to the CRC, CRIOAc can have immediate advice in order to easily participate in national or international clinical trials and galvanise our projects.
International Centre for Infectiology Research
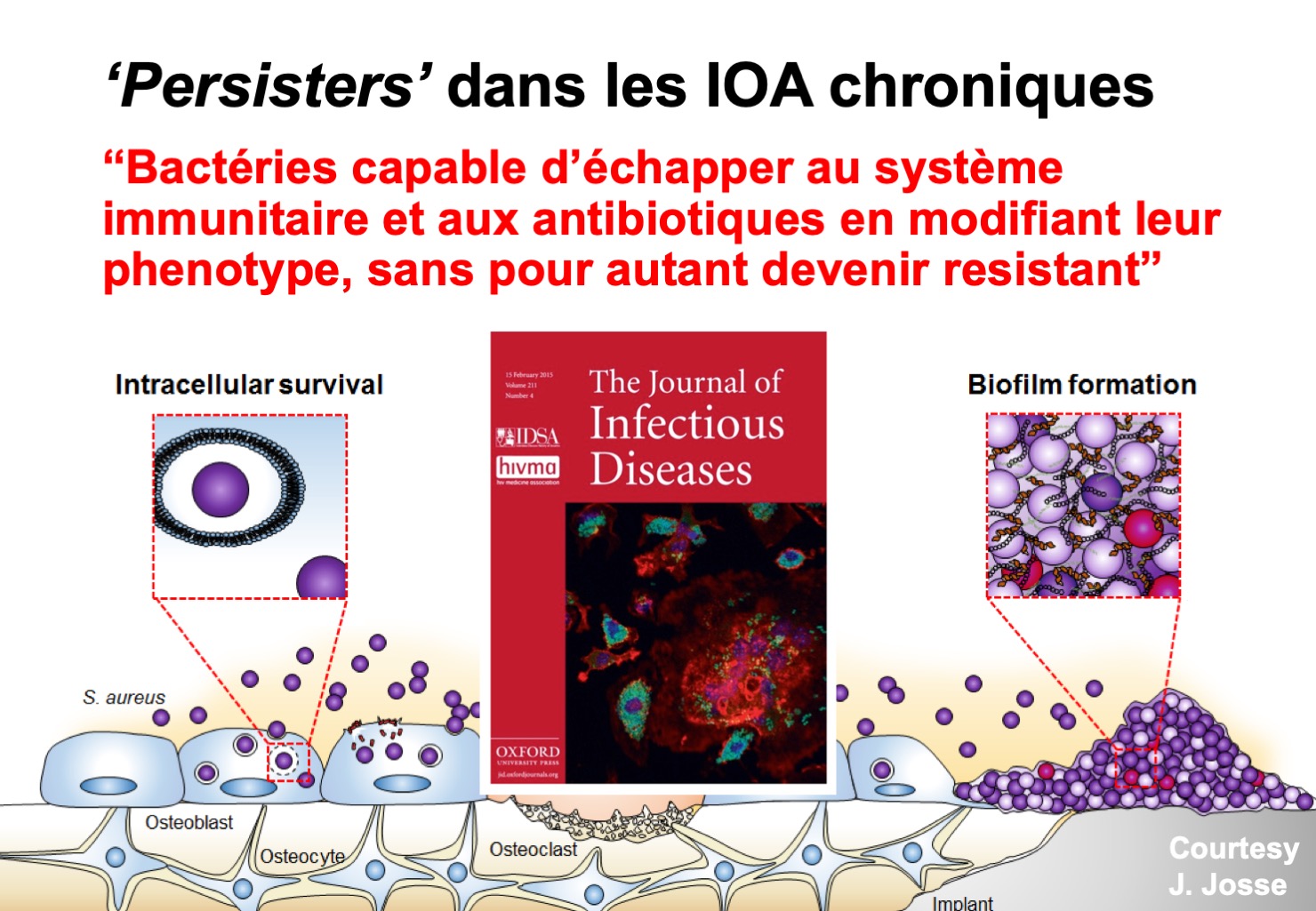
Much progress is needed to better understand the pathophysiology of BJI, especially regarding the mechanisms of bacterial persistence and immune system evasion. CRIOAc Lyon has developed, at the INSERM 1111 unit of the International Centre of Infectiology Research CIRI (team studying the pathogenesis of staph infections, Pr. F. Vandenesch) an entire line of research on the mechanisms of persistence of staphylococci, which are the main germs responsible for BJI. Biofilm production is a mechanism used by bacteria to persist in contact with an implant. Biofilm can be microscopic, or can be seen with the naked eye, as in the figure opposite. When biofilm is formed, it is inseparable from the implant. Finding the reasons for this phenomenon is essential to getting closer to a cure.

Biofilm visible to the naked eye in chronic staphylococcal infection of a knee prosthesis
We have developed cell cultures of osteoblasts and osteoclasts that we infect in vitro with different strains of staphylococci.
All these models allow us to better understand the pathophysiology of BJI, and in particular the mechanisms of bacterial persistence. Many bacteria are able to change their phenotype and become “persisters” making their eradication difficult or even impossible. All this work allows us to publish articles in international medical and scientific journals.
Support CRIOAc Lyon research
You can participate directly and actively in CRIOAc Lyon research by making a donation to support our projects via Fondation HCL by clicking HERE.






